
Information regarding tissue prep and sample submission for Visium, GeoMx DSP, Curio Seeker, and Xenium processing.
| Platform | Visium | Xenium | GeoMx DSP | Curio Seeker | CurioTrekker |
| Sample Type | Fresh Frozen, Fixed Frozen, or FFPE | Fresh Frozen, Fixed Frozen, or FFPE | Fresh Frozen, Fixed Frozen, or FFPE | Fresh Frozen | Fresh Frozen |
| Submission Format | blocks, glass slides, or 10x custom slides | blocks or 10x custom slides | blocks or standard slides (freshly cut) | blocks | blocks |
| Recommended QC | Manual FF RIN >7; CytAssist or HD DV200 > 30% | none | none | RIN > 7 | RIN > 7 |
| Size | Manual FF: 6.5 mm x 6.5 mm (4 per slide) CytAssist: 6.5 mm x 6.5 mm or 11mm x 11mm (2 per slide); HD: 6.5 mm x 6.5 mm | 10.45 mm x 22.45 mm | up to 40 x 17 mm | 3 mm x 3mm tile; 10 mm x 10mm tile | 10 mm x 10mm tile |
| Species | Manual: diverse; CytAssist or HD: human or mouse | diverse | human or mouse | diverse | diverse |
| Analyte | Manual FF: RNA, CytAssist: RNA and protein; HD: RNA | predesigned or custom targeted RNA panels | RNA or protein | RNA | RNA or ATAC |
| Resolution | Manual or CytAssist: 55um spots; HD: 8um bins (can be parsed into 2um) | subcellular | minimum 50-100 cells per ROI | 10 um beads, 1-2 cells/bead | single nuclei |
| Visualization | H&E or immunofluorescence | H&E or immunofluorescence (post Xenium processing) | four-color fluorescence | serial section required | serial section required |
| Deliverables | SpaceRanger analysis pipeline output | Xenium pipeline output | Quality Control Report with R objects | Curio pipeline output | Curio pipeline output |
| Downstream Analysis | Loupe Browser Visualization Software and 3rd party tools (e.g. Seurat) | Xenium Browser Visualization Software and 3rd party tools (e.g. Seurat) | vendor supplied R packages | Compatible with 3rd party tools (e.g. Seurat) | Compatible with 3rd party tools (e.g. Seurat) |
The Visium CytAssist workflow follows a standard histological workflow: sectioning, tissue preparation, staining (H&E or IF), and imaging all take place on a standard glass slide. Additional tissue preparation is followed by probe hybridization on the same glass slide. During this phase, probes hybridize to approximately 18,000 genes, or RNA targets, within the tissue section, offering whole transcriptome gene expression profiling.
Next, the CytAssist instrument incorporates two normal glass slides and a Visium slide with a pair of Capture Areas. This arrangement allows for the alignment of tissue sections on the standard slides with the Capture Areas.
The instrument uses a brightfield image for spatial referencing during data analysis, then the transcriptomic probes from the tissue hybridize onto the Visium slide. The steps that follow, starting with probe extension, adhere to the standard Visium procedure outside of the instrument.


CytAssist Slide Configuration
A Visium Gene Expression (GEX) slide is comprised of two or four tissue capture areas. A standard capture area is 6.5mm x 6.5mm and contains ~5000 barcoded spots measuring 55 µm in diameter and spaced so that the distance between the centers of each spot is 100 µm. The XL capture area is 11mm x 11mm and contains ~14000 barcoded spots.
Depending on the tissue, an average of 1-10 cells will cover a spot. The spatial barcode assigned to the spot is incorporated during cDNA synthesis and enables gene expression data to be mapped back to its location within the tissue. Data is processed with the 10X SpaceRanger analysis software and can be visualized with the Loupe Browser software.

CytAssist Workflow – Investigators section tissue onto plain glass slides. Once tissue is placed on the slide, the slide is transported to the AGC. AGC staff will perform H&E staining, imaging, library prep, sequencing, and SpaceRanger data processing.
Tissue Submission – Tissue processed following the recommended protocols can be submitted directly to the AGC for sectioning, staining/imaging, library prep, sequencing, and data processing. The cryomold used for embedding should be of appropriate size to fit the tissue sample. Preferred size for cryoblock submission is 10 mm.
Sample Requirements
- Species Specific – only human and mouse samples currently supported
- Fresh/Frozen, Fixed/Frozen, or FFPE
- Ensure desired tissue area will fit within the 6.5 x 6.5 mm or 11 x 11 mm capture area
- Section tissue within the allowable target area on the plain glass slide – see page 6 of the 10x Genomics recommended tissue prep guide
- Recommended section thickness is 3-10 µm
- RNA quality of the block should be assessed prior to sectioning. The DV200 should be ≥ 30%.
Visium CytAssist Protein Expression Protein Panel – 35-plex CytAssist Panel Antibodies
- Targets intracellular and extracellular antigens
- 4 isotype control (for data normalization)
A description of the panel composition can be found here.
Visium HD use the CytAssist workflow. HD slides contain two 6.5 x 6.5 mm Capture Areas with a continuous lawn of oligonucleotides arrayed in millions of 2 x 2 µm barcoded squares without gaps, achieving single cell–scale spatial resolution. The data is output at 2 µm, as well as multiple bin sizes. The 8 x 8 µm bin is the recommended starting point for visualization and analysis.

CytAssist Workflow – Investigators section tissue onto plain glass slides. Once tissue is placed on the slide, the slide is transported to the AGC. AGC staff will perform H&E staining, imaging, library prep, sequencing, and SpaceRanger data processing.
Tissue Submission – Tissue processed following the recommended protocols can be submitted directly to the AGC for sectioning, staining/imaging, library prep, sequencing, and data processing. The cryomold used for embedding should be of appropriate size to fit the tissue sample. Preferred size for cryoblock submission is 10 mm.
Sample Requirements
- Species Specific – only human and mouse samples currently supported
- Fresh/Frozen, Fixed/Frozen, or FFPE
- Ensure desired tissue area will fit within the 6.5 x 6.5 mm capture area
- Section tissue within the allowable target area on the plain glass slide – see page 6 of the 10x Genomics recommended tissue prep guide
- Recommended section thickness is 3-10 µm
- RNA quality of the block should be assessed prior to sectioning. The DV200 should be ≥ 30%.
Fresh/Frozen tissue (Manual Visium Assay)
Visium Gene Expression is essentially a set of slides, reagents, and software tools that enable a person to do whole transcriptome analysis on a tissue section. Thus, there are a variety of ways you can opt to use the platform and work with the Advanced Genomics Core.
Visium Slide Submission – Investigators section tissue onto the Visium slide. Once tissue is placed on the slide, the slide is stable at for up to four weeks so it can be easily transported on dry ice to the AGC. AGC staff will perform H&E staining, imaging, library prep, sequencing, and SpaceRanger data processing.
Work with one of the tissue processing cores on campus – Investigators can coordinate with their preferred tissue facility to section onto the Visium slide. You can also work directly with that or another facility to stain and image the slide (required if performing IF), but you will need to meet the 10X imaging requirements. This option needs to be tightly coordinated with the AGC for hand-off of the slides. There is not a safe stopping point so AGC staff need to be prepared to accept imaged slides and proceed with the workflow.
Tissue Submission – Tissue processed following the recommended protocols can be submitted directly to the AGC for sectioning, staining/imaging, library prep, sequencing, and data processing. The cryomold used for embedding should be of appropriate size to fit the tissue sample. Preferred size for cryoblock submission is 10 mm.
Self Service – The Visium platform does not require specialized instrumentation. Reagents can be purchased directly from 10x Genomics and users can prep samples in their own lab space. These samples are submitted to the AGC as user-made libraries ready for Next Generation Sequencing.
Sample Requirements
- Freshly obtained tissue should be snap frozen to prevent RNA degradation
- The recommended freezing method uses an isopentane and liquid nitrogen bath
- Frozen tissue should be embedded in Optimal Cutting Temperature (OCT), a freezing and embedding compound (can be done simultaneously with freezing process)
- RNA quality of the tissue block should be assessed prior to sectioning. The RNA Integrity Number (RIN) should be ≥ 7
- Each new tissue type for a lab is required to undergo a tissue optimization step so that the correct permeabilization time can be established prior to scheduling a Visium GEX appointment
Helpful Documents
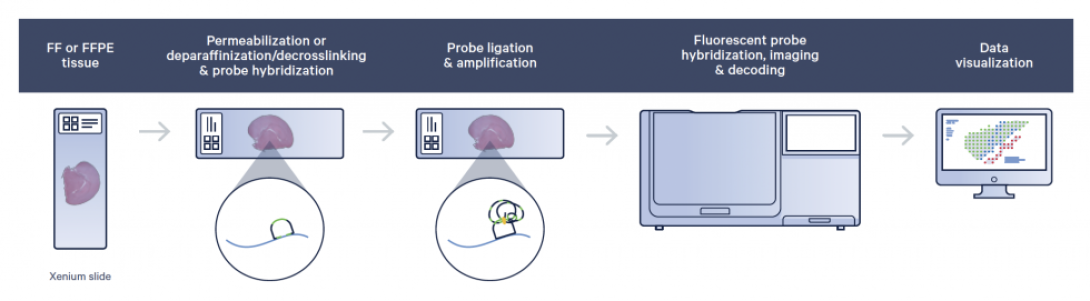
FF or FFPE tissue is sectioned onto a Xenium slide (each slide has an imageable area of 12mm x 24mm). Sections are treated to access the RNA for labeling with circularizable DNA probes. Probe ligation generates a circular DNA probe which is enzymatically amplified. Slides are placed in the Xenium Analyzer where the sample undergoes successive rounds of fluorescent probe hybridization, imaging, and removal; creating bright, easy to image signal with a high signal-to-noise ratio.
An optical signature specific to each gene is generated, enabling target gene identification. Finally, a spatial map of the transcripts is built across the entire tissue section. Data is processed with the 10x Xenium analysis software and can be visualized with the Xenium Browser software.

- Fresh/Frozen, Fixed/Frozen or FFPE
- Section Thickness
- 5 µm (FFPE)
- 10 µm (Fresh/Frozen; Fixed Frozen)
- Tissue processed following the recommended protocols can be submitted directly to the AGC for sectioning and Xenium processing.
- Investigators section tissue onto the Xenium slide. Once tissue is placed on the slide, the slide is stable at for up to four weeks so it can be easily transported to the AGC.
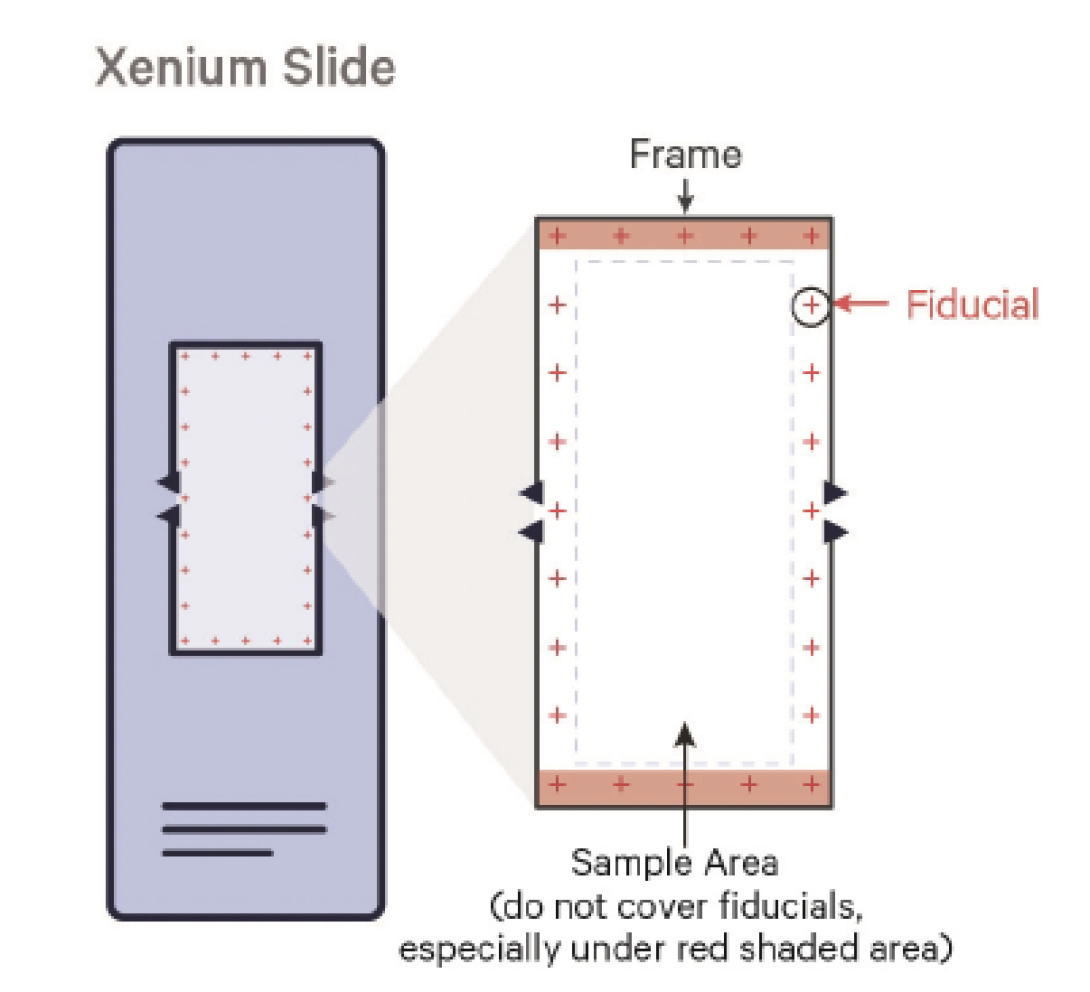
Xenium slides have an imageable area outlined by a fiducial frame measuring 12 mm x 24 mm. Specimens need to fit within the available sample positioning area measuring 235 mm2 (10.45 mm x 22.45 mm).
Available now:
- Human Brain
- Human Breast
- Human Colon
- Human Immuno-Oncology
- Human Lung
- Human Multi-Tissue and Cancer
- Human Skin
- 5K Human Pan Tissue & Pathways Panel
- Mouse Brain
- Mouse Tissue Atlas
- 5K Mouse Pan Tissue & Pathways Panel
- Fully Custom – up to 480 genes
note – up to 100 custom targets can be added to pre-designed panels
The Xenium with Cell Segmentation Staining method uses a specialized stain mix to precisely identify cell boundaries. These labels facilitate automatic cell segmentation, providing clear boundary definition when membrane signals are pronounced and alternative strategies for boundary identification when membrane staining is unclear. The Xenium Multi-Tissue Stain Mix includes four labeling agents targeting cell membranes, cytoplasm, ribosomal RNA, and nuclei with DAPI.
Cell Segmentation Antibodies
- Membrane markers: E-cadherin (Cdh1), ATPase1a1, CD45
- Cytoplasmic RNA: 18s
- Cytoplasmic proteins: vimentin, aSMA
Cell Segmentation is automatically included with 5K Pan Tissue & Pathways Panels and can be added to any other Xenium pre-designed or custom panel by request.
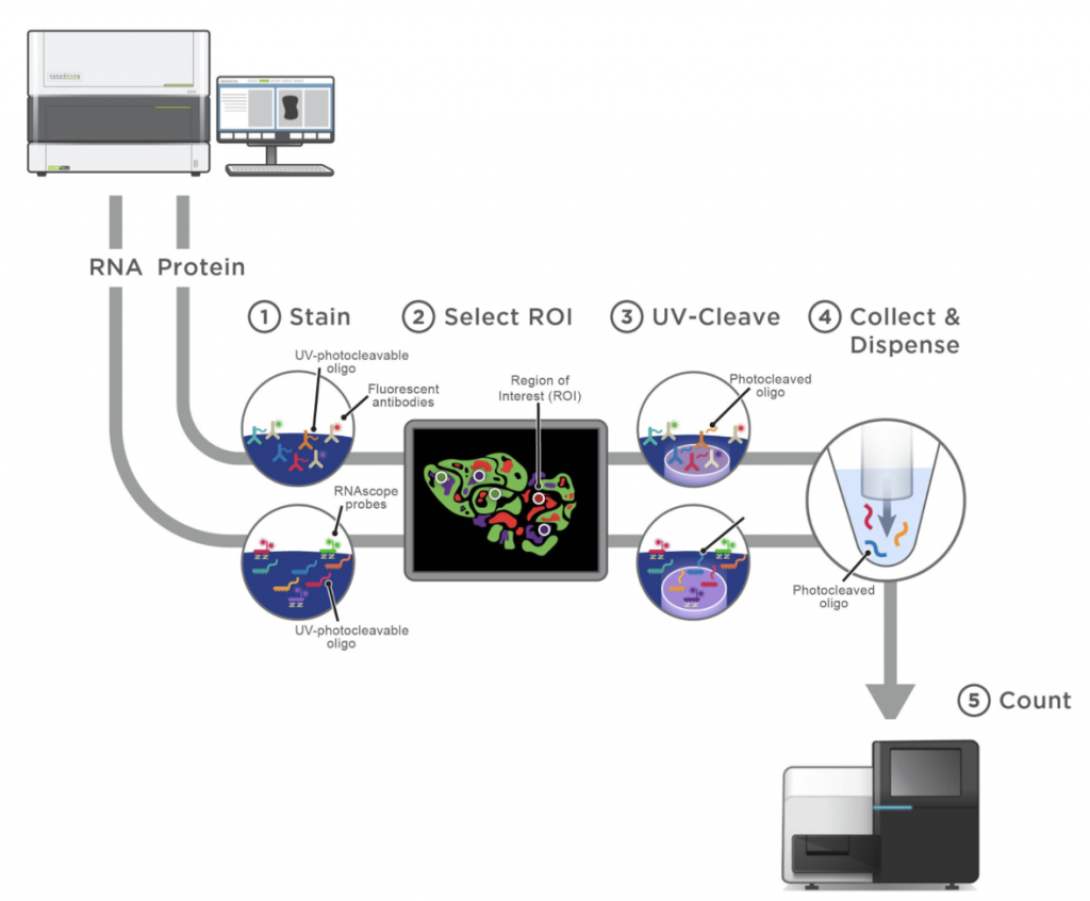
NanoString’s GeoMx Digital Spatial Profiler (DSP) combines spatial and molecular profiling technologies by generating digital whole transcriptome or protein expression data for up to 4 tissue slides per day. A series of imaging reagents tagged with fluorophore are used to visualize the tissue and a series of oligo-tagged profiling reagents are used to interrogate protein or transcript expression. Once regions of interest (ROIs) are selected by the investigator, DNA oligos bound to the profiling reagents are sequentially released, collected into individual wells on a 96-well plate, and subjected to Illumina library prep. Expressions levels are readout using an Illumina Sequencer and analyzed using the DSP interactive software.

- Whole Transcriptome Atlas (human or mouse)
- Cancer Transcriptome Atals (human)
- Protein Assays with NGS readout (human or mouse)
- FFPE or Fresh/Frozen tissue
- Species Specific – only human and mouse samples currently supported
- Recommended section thickness is 5 µm
- Tissue should be sectioned onto positively charged slides no more than 2 weeks prior to your Spatial Assay Appointment
- A test slide for a scanning only run (without ROI definition, UV illumination, or ROI collection) is strongly recommended to expedite ROI selection the day of the experiment and validate the fluorescent staining of your morphology markers. All requests utilizing custom antibodies are required to undergo this process.
AGC GeoMx DSP Reagents June 2021 – present
| Vendor | Part Number | Description |
| nanoString | GeoMx NGS RNA CTA Hs | GeoMx Cancer Transcriptome Atlas |
| nanoString | GeoMx NGS RNA WTA Hs | GeoMx Human Whole Transcriptome Atlas |
| nanoString | GeoMx NGS RNA WTA Mm | GeoMx Mouse Whole Transcriptome Atlas |
| nanoString | various | Protein Module for NGS |
The nanoString GeoMx DSP platform is highly interactive and projects require extensive planning. Schedule a consultation here. Once your assay has been selected, additional key decision points are:
Slides and Samples: Multiple sections or sections from multiple tissues can be placed on the same slide for DSP processing. The only caveat is that all sections must fit within the GeoMx gasket. The gasket dimensions are shown here.
Recommended slides:
- Leica Bond Plus (Cat# S21.2113.A)
- Fisherbrand Superfrost Plus (Cat # 12-550-15)
Morphology Marker Selection: Morphology markers are employed to guide selection of ROIs based on visual staining patterns. While we stock nanoString morphology kits that contain common tissue markers, the DSP system is agnostic to the imaging reagents. We have a list of antibodies previously validated by the nanoString Technology Access Program. Custom antibodies are also supported as long as the antibody conjugate works with one of the 4 channels on the instrument. One of the fluorescent channels needs to be reserved for a nuclear stain, leaving three channels open for antibodies of interest. The list of available fluorescent channels are:
| Channel | Excitation Peak/Bandwidth | Emission Peak/Bandwidth |
| FITC / 525 nm | 480 / 28 nm | 516 / 23 nm |
| Cy3 / 568 nm | 538 / 19 nm | 564 / 15 nm |
| Texas Red / 615 nm | 588 / 19 nm | 623 / 30 nm |
| Cy5 / 666 nm | 645 / 19 nm | 683 / 30 nm |
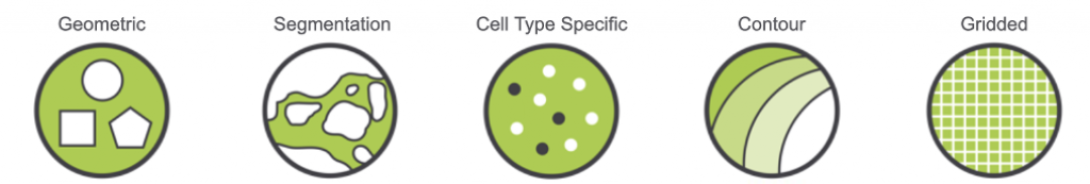
Defining Regions of Interest: There are multiple approaches for defining ROIs, including placing geometric shapes around specific areas (Geometric), drawing concentric circles surrounding a histological landmark (Contour), and gridding across the entire slide (Gridded).
You can select 1-96 ROIs per slide as long as they do not overlap with each other. ROI size determines total counts collected. The largest allowable ROI is ~700 μm. While the smallest region for illumination is 10 μm, this is insufficient for generating profiling data. Ultimately, the minimum ROI size will depend on your tissue, the types of cells within your ROIs, and, for RNA, the transcriptional activity of the cells. We do not recommend starting with fewer than 50 cells per ROI (non-segmented). If segmenting, then larger ROIs will be required.
Regions can also be profiled using immunofluorescence/RNAscope patterns (segmentation, cell type specific). These are defined by combinations of the presence and absence of specific morphology markers will become areas of illumination (AOIs) during slide processing. Each AOI gets collected separately, thereby creating two or more expression profiles for the same ROI.
Considerations when creating segment masks include:
- Segments cannot overlap or extend outside an ROI
- Segments should be as contiguous as possible, with clean margins separating each segment from off-target tissue
- Segments composed of numerous small particles will have reduced fidelity and higher background from adjacent tissue
Defining ROIs and their segmentation pattern is a lengthy process and requires a significant time commitment on the investigators part the day of the experiment. All first time users are required to come to NCRC for ROI selection.
Data Analysis: The AGC will complete the nanoString NGS pipeline to generate the digital count (DCC) files and notify you when they have been loaded onto the DSP. To perform data analysis, please reserve time on the instrument using our nanoString GeoMx Data Analysis calendar in MiCores. Please note days/times the instrument is available for analysis is limited based on projects the AGC is processing on the device.
Helpful Documents
Useful Links
- Abcam – NanoString collaboration to offer GeoMx™-formatted antibodies
- ImageJ – image analysis software
- nanoString Academy – DSP training courses
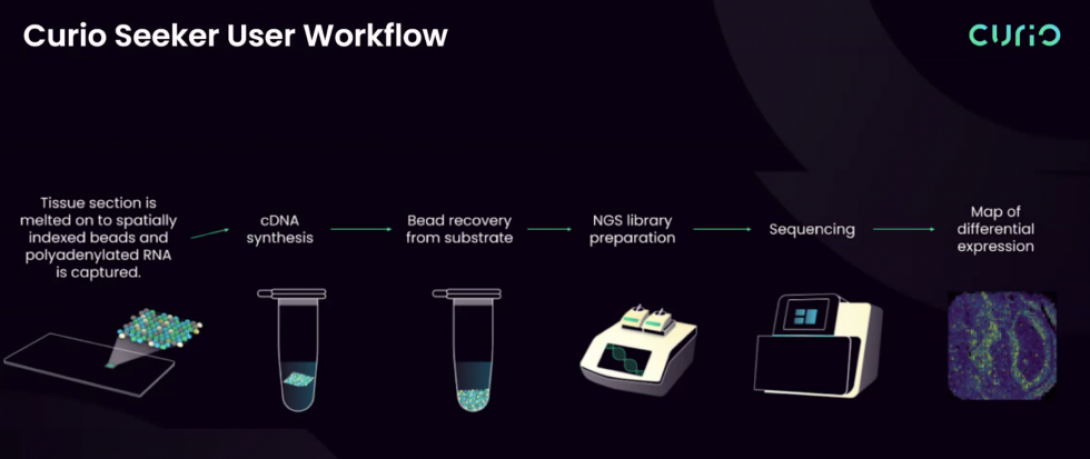
Curio Seeker tiles are 3 x 3 mm or 10 x 10 mm and contain ~10 µm barcoded beads. The spatial barcode assigned to the bead is incorporated during cDNA synthesis and enables gene expression data to be mapped back to its location within the tissue. Data is processed with the Curio Bioscience analysis software.
Fresh/Frozen tissue
- Freshly obtained tissue should be snap frozen to prevent RNA degradation
- The recommended freezing method uses an isopentane and liquid nitrogen bath
- Frozen tissue should be embedded in Optimal Cutting Temperature (OCT), which can be done simultaneously with freezing process
- RNA quality of the tissue block should be assessed prior to sectioning. The RNA Integrity Number (RIN) should be ≥ 7
Curio Seeker is a set of slides, reagents, and software tools that enable a person to do whole transcriptome analysis on a tissue section. Thus, there are a variety of ways you can opt to use the platform and work with the Advanced Genomics Core.
Tissue Submission – Tissue processed following the recommended protocols can be submitted directly to the AGC for sectioning, staining/imaging, library prep, sequencing, and data processing. The cryomold used for embedding should be of appropriate size to fit the tissue sample. Preferred size for cryoblock submission is 10 mm.
cDNA Submission – Investigators section process onto the tile, perform the Curio assay, and submits the recovered cDNA to AGC. AGC staff will perform library prep, sequencing, and Curio Bioscience pipeline data processing.
Self Service – The Curio Seeker platform does not require specialized instrumentation. Reagents can be purchased directly from Curio Bioscience and users can prep samples in their own lab space. These samples are submitted to the AGC as user-made libraries ready for Next Generation Sequencing.
Curio Trekker's Slide-Tags Methodology
The Curio Trekker Platform employs the innovative Slide-Tags approach, capturing the rich spatial transcriptomics data from intact tissue sections. By tagging single nuclei with spatial barcode oligonucleotides tied to DNA-barcoded beads, Curio Trekker offers precise spatial resolution for single-nucleus profiling.
Key Features:
- Spatial Barcode Technology: Assigns spatial coordinates to individual nuclei via DNA barcode tags, seamlessly integrating with 10x Genomics 3' Gene Expression and Multiome assays.
- Flexible Assay Integration: Versatility in assay selection facilitates comprehensive single-nucleus profiling, encompassing gene expression and chromatin accessibility, while preserving spatial context.
- Species Agnosticism: Curio Trekker's capabilities are not limited by species, providing a universal tool for diverse research applications.
- Tissue Compatibility: Optimized for fresh frozen, OCT-embedded tissues, ensuring the structural integrity and quality of samples.
- Holistic Imaging: Alignment with histological imaging on serial tissue sections offers a complete view of tissue morphology alongside molecular data.
Curio Trekker is the only platform available at the AGC that allows simultaneous capture of both gene expression and chromatin accessibility data from the same sample, maintaining the spatial information critical for comprehensive biological insights.
University of Michigan
2800 Plymouth Rd.
Ann Arbor, MI 48109-2800